Precautions Against Mine Gas |
|
| For
every ton of coal removed from the working face, anything
up to 200 m3 (7000 ft3) of mine gas is released; the average
amount in the Ruhr coal-mining district is 30 m3 (about 1000
ft3). This gas consists of hydrocarbons, chiefly methane (CH4)
and is known as firedamp, more particularly when it occurs
in an explosive mixture with air (when the gas concentration
in the air is between 4.5 and 14.5%). Methane is a colorless,
odorless, and tasteless gas.
|
|
| Firedamp
explosions have been the cause of many catastrophes and a
vast number of deaths in the history of coal mining. The initial
explosion is liable to ignite the cloud of coal dust that
the blast disperses. When the air contains between 700 and
1000 grams of coal dust per cubic meter, it constitutes an
explosive mixture. A coal-dust explosion produces carbon monoxide
(CO), which is a serious danger to human life because of its
toxicity.
|
|
 |
|
| Accordingly
the main object of research, supervision and legislation relating
to safety in coal mines is to develop and improve the precautions
against the occurrence of such mishaps. Essentially, the aim
is to eliminate all possibility of ignition of such explosive
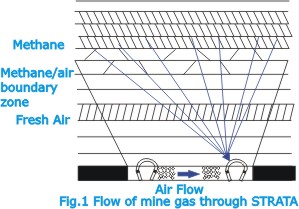
mixtures. The emission of gas from the coal is a natural phenomenon
associated with the constitution of the coal and adjacent
strata (Fig.1), and except in some rare circumstances where
methane gas can be removed in advance by suction, it is not
possible to reduce or effectively control this emission.
|
|
| The
application of special procedures in extracting the coal,
high speed of advance at the coal face, complete sealing off
of old workings by suitable stowing (backfilling), etc., may
have a favorable effect. It nevertheless remains essential
to conform closely to safety regulations and to take all manner
of technical precautions to prevent the occurrence of any
spark or flame that might set off an explosion, including
a strict ban on smoking. There remain potential sources of
ignition in the use of explosives and electricity underground.
Frictional heating of machinery and spontaneous-combustion
phenomena, which may arise under certain conditions are also
hazards.
|
|
| An
effective measure consists in keeping the workings supplied
with fresh air in quantities large enough to ensure that the
mine-gas concentration will at all times remain below the
explosive level. The first important development in overcoming
the firedamp menace was the invention of the safety lamp by
Davey in 1816.
|
|
| |
|
 |
|
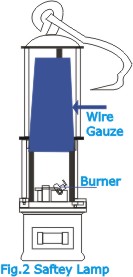
| The
lamp which burns a liquid fuel is provided with an enclosure
of metal-wire gauze above the glass (Fig.2). If the air surrounding
the lamp contains mine gas, the flame of the lamp will ignite
the gas, but the latter will burn only inside the gauze enclosure;
the flame will not ignite the gas all round the lamp. It is
the high thermal conductivity of the gauze that arrests flame
propagation. |
|
 |
|
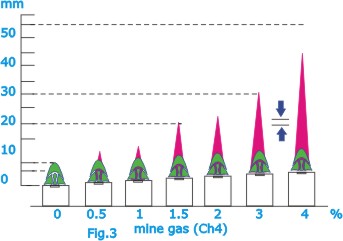
| The
lamp is used today as a detector for gas, which burns with
a characteristic flame called a gas cap that appears when
the flame in the lamp is lowered. The length of the gas cap
provides an indication of the percentage of gas in the air
(Fig.3). A countermeasure against the formation of highly
inflammable and therefore explosive coal-dust clouds consist
in dusting i.e., specified quantities of stone dust are deposited
throughout the mine, more particularly just before blasting
is to take place. |
|
 |
|
|
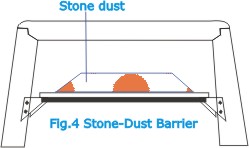
The
cloud of dust thrown up by the explosion is rendered nonflammable
by the presence of the stone dust. Stone dust is also used
as a means of arresting the propagation of explosions. For
this purpose a device called a stone-dust barrier (Fig.4)
is installed at strategic points in the mine. It may take
the form of a light tilting platform on which a quantity of
stone dust is placed. In the event of an explosion in the
vicinity, the dust is flung off and forms a dense cloud which
absorbs some of the energy of the blast and also exercises
a cooling action which smothers the flame. |
|
|
o
DISCLAIMER o
CONTACT US |