Sulphuric Acid |
|
|
Although
sulfuric acid is now one of the most commonly used chemicals,
it was probably little known before the 16th cent. It was
prepared by Johann Van Helmont (c.1600) by destructive distillation
of green vitriol (ferrous sulfate) and by burning sulfur.
The first major industrial demand for sulfuric acid was the
Leblanc process for making sodium carbonate (developed c.1790).
Sulfuric acid was produced at Nordhausen from green vitriol
but was expensive. |
|
| A
process for its synthesis by burning sulfur with saltpeter
(potassium nitrate) was first used by Johann Glauber in the
17th cent. and developed commercially by Joshua Ward in England
c.1740. It was soon superseded by the lead chamber process,
invented by John Roebuck in 1746 and since improved by many
others. The contact process was originally developed c.1830
by Peregrine Phillips in England; it was little used until
a need for concentrated acid arose, particularly for the manufacture
of synthetic organic dyes. |
|
 |
|
| In
the pure state, sulphuric acid is a clear, colorless, oily
liquid. One-hundred percent H2SO4 has its melting point at
10°C; when heated, it gives off SO3 until the concentration
of the acid has fallen to 98.5%, and it then boils at a constant
temperature of 338°C. Considerable evolution of heat occurs
when concentrated sulphuric acid is diluted with water. Substantial
amounts of SO3 can dissolve in the acid. The resulting solution
is known commercially as fuming sulphuric acid.
|
|
| Sulphuric
acid does not occur as a free acid in nature. It is found
only in the form of its salts (sulphates): gypsum (CaSO4.2H2O),
Epsom salts (MgSO4.7H2O), barite (BaSO4) and Glauber’s
salt (Na2SO4.1OH2O). Up to about the eighteenth century, sulphuric
acid was made by heating alum (aluminum potassium sulphate)
or iron vitriol (hydrous ferrous sulphate).
|
|
| This
method was superseded by the burning of natural sulphur with
saltpeter, which eventually evolved into the so-called lead-chamber
process, which has the advantage that the acid can be obtained
in any desired concentration, whereas the highest attainable
concentration with the lead chamber process is 78%.
|
|
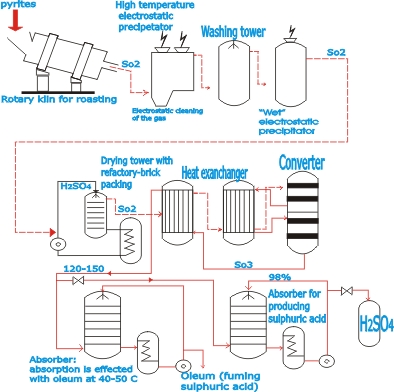
| The
contact process is as follows, Sulphur dioxide (SO2) is obtained
by roasting iron pyrites (FeS2) in a rotary kiln, shelved
roasting kiln or fluidized bed kiln. Which of these kiln types
is employed depends on the particle size and nature of the
pyrites to be processed. When the gases from the roasting
process have cooled in gas ducts, by radiation of heat from
1000 oC to about 400°C -500°C, the dust they contain is
removed in electrostatic precipitators (electric fitters).
|
|
| |
|
| Next,
the SO2 gas is passed through a washing tower, where constituents
that are present in vapor form mainly compounds of arsenic,
selenium and chlorine are removed with sulphuric acid serving
as the washing liquid. Remaining traces of impurities present
as very fine suspended droplets (fog) are removed in an irrigated
electrostatic precipitator (wet Precipitator). Then the gas
is dried by being brought into contact with concentrated (98%)
sulphuric acid.
|
|
| A
blower draws in the cold dried SO2 gas and delivers it into
the converter, which is a tank or tower in which a suitable
catalyst e.g., vanadium pentoxide (V2O5) is placed in layers
on shelves or arranged in some other appropriate manner to
ensure through contact with the gas. The reaction whereby
SO2 is converted to SO3 by oxidation (2SO2+O2-> 2SO3) takes
place at 430°C to 550°C. A heat exchanger installed before
the converter serves to cool the gas discharged from the converter
and at the same time preheats the incoming gas flowing to
the converter.
|
|
Sulfuric
acid is one of the most important industrial chemicals. More
of it is made each year than is made of any other manufactured
chemical; more than 40 million tons of it were produced in
the United States in 1990. It has widely combined uses and
plays some part in the production of nearly all manufactured
goods.
|
|
The
major use of sulfuric acid is in the production of fertilizers,
e.g., superphosphate of lime and ammonium sulfate. It is widely
used in the manufacture of chemicals, e.g., in making hydrochloric
acid, nitric acid, sulfate salts, synthetic detergents, dyes
and pigments, explosives, and drugs.
|
|
| It
is used in petroleum refining to wash impurities out of gasoline
and other refinery products. Sulfuric acid is used in processing
metals, e.g., in pickling (cleaning) iron and steel before
plating them with tin or zinc. Rayon is made with sulfuric
acid. It serves as the electrolyte in the lead-acid storage
battery commonly used in motor vehicles acid for this use,
containing about 33% H2SO4 and with specific gravity about
1.25, is often called battery acid.
|
|
|
o
DISCLAIMER o
CONTACT US |